Benefit from Mecmesin's 40 years' experience as a leader in developing testing solutions

Scaling manufacturing while maintaining quality
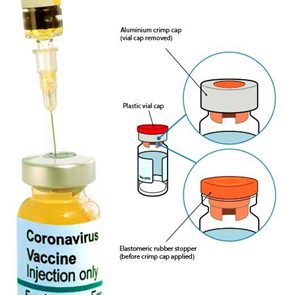
COVID-19 vaccines, as parenteral medications, enter the body by injection through the tissue and circulatory system. In line with most parenteral drug products, these vaccines are manufactured in glass vials (or phials) and sealed with an elastomeric rubber stopper and a crimp cap.
Testing the physical properties of these vital drug container systems is a key factor in quality-control testing.
Vial sealing is a critical process step during ‘fill and finish’ operations, as it defines the seal quality of the final product, ensuring sterility and preventing leakage. Of particular interest is the effectiveness of the rubber stopper when in its compressed state and how well it is restrained by the aluminium cap, which itself is folded and crimped securely under the vial flange.
The Residual Seal Force (RSF) is commonly measured, as part of a ‘Container Closure Integrity (CCI)’ testing process, to characterise the performance of capping equipment and is referenced in USP 1207.3.
A universal testing machine applies force onto the sealing surface.
The rubber stopper acts like a compressed spring, pushing up against the test system. At the very moment that the tester overcomes this compressive resistance there is a discernible kink in the Force/Displacement curve, which equates to the RSF.
The Torque Retention of the aluminium crimp cap is also a good indicator of seal quality and is significantly impacted by the Residual Seal Force. The vial is held in the jaws of a torque tester and the crimp cap is rotated to establish whether it breaks loose. Crimp caps which break loose at low torques may not be adequately restrained by the rubber stopper and therefore correlate to low RSF values.